Methods of Separating Mixtures and purifying substances
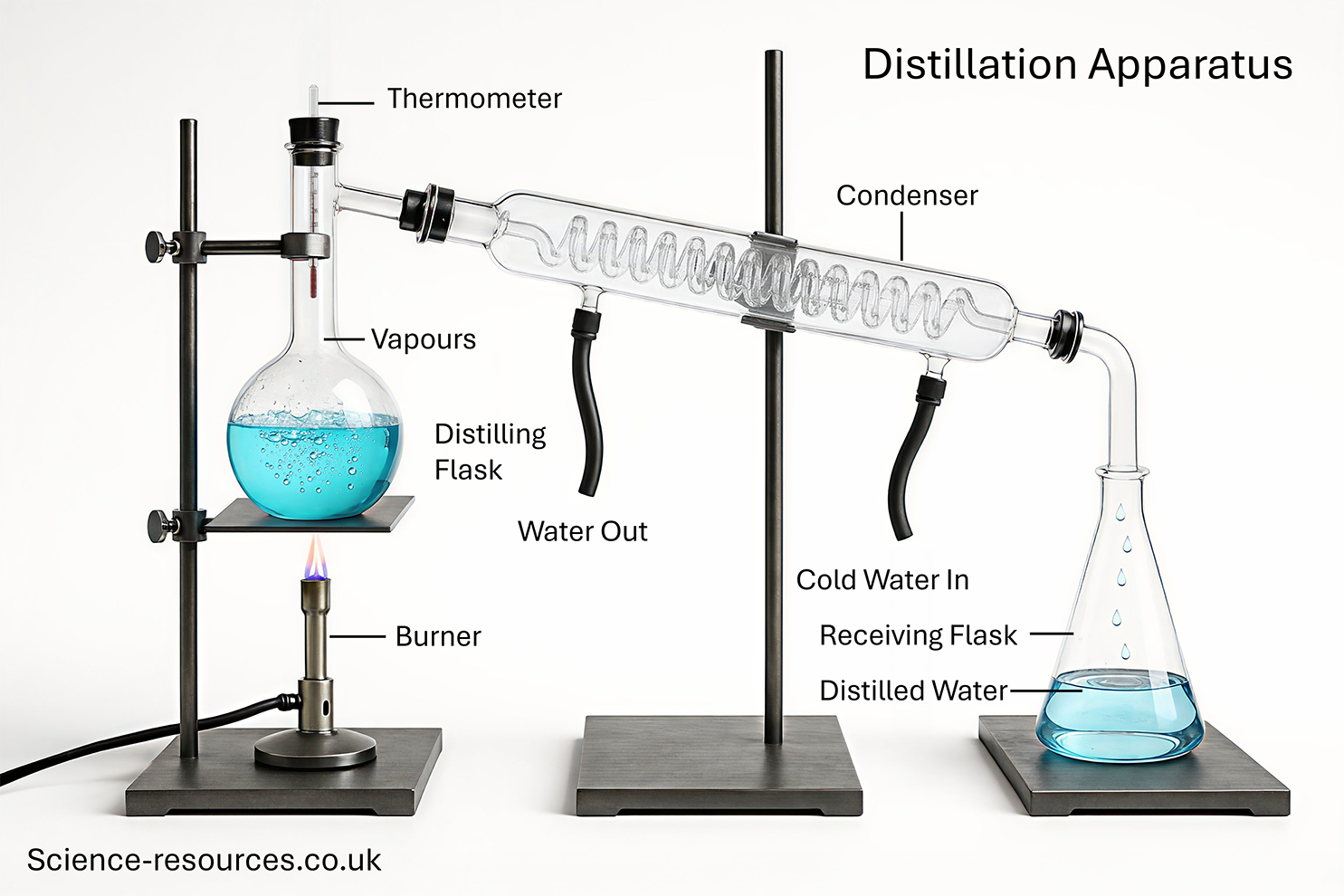
Rock salt is a mixture of salt and sand. Salt and sand are both compounds but because the salt is soluble in water and the sand isn't, the two can be separated using the following methods: Simple distillation is used to separate a mixture of a liquid and a solid, for example ink. (1) The liquid is heated and boils off. (2) The vapour is cooled by cold water, condensed and collected leaving the solid behind. This can be used to purify sea water or tap water. Distillation experiment Filtration and Evaporation
Simple
Distillation
Fractional distillation is used for separating a mixture of liquids. (1) The mixture of liquids are heated and each boil off at their own temperature. (2) The vapour passes up through a fractionating column. (3) The vapour is cooled by cold water, condensed and collected. Fractional distillation is used on a large scale to separate the components of crude oil, because the different hydrocarbons have different boiling points (Distillation of crude oil). Fractional
Distillation
Test your understanding of the different methods used to separate mixtures in the laboratory. 1. Which method would be most suitable for separating sand from a mixture of sand and water? 2. In evaporation, what is left behind in the evaporating dish after the liquid has turned into a gas? 3. Which technique relies on substances having different boiling points to separate them? 4. Chromatography separates substances like ink dyes based on which property? 5. Which technique would you use to separate two liquids that do not mix (immiscible), such as oil and water? 🧪 Knowledge Check: Separation Techniques
Click to Reveal Answers
2. The solid solute (The liquid evaporates, leaving the solid behind).
3. Distillation (Boiling points determine which liquid evaporates first).
4. Solubility in a solvent (Different dyes travel different distances).
5. Decanting or using a separating funnel (Used for liquids that naturally separate into layers).