Symbols and formulae
Elements and symbols Elements Symbols Elements are shown by letters from the alphabet. These letters are also called symbols. The symbol for an element is either a single capital letter or a capital letter with a lower case letter after it. The periodic table
An element is a substance that is made of only one kind of atom, and cannot be split into simpler substances. Some examples of elements are oxygen, hydrogen, gold, and iron.
For example:
There are 118 chemical elements listed in the periodic table: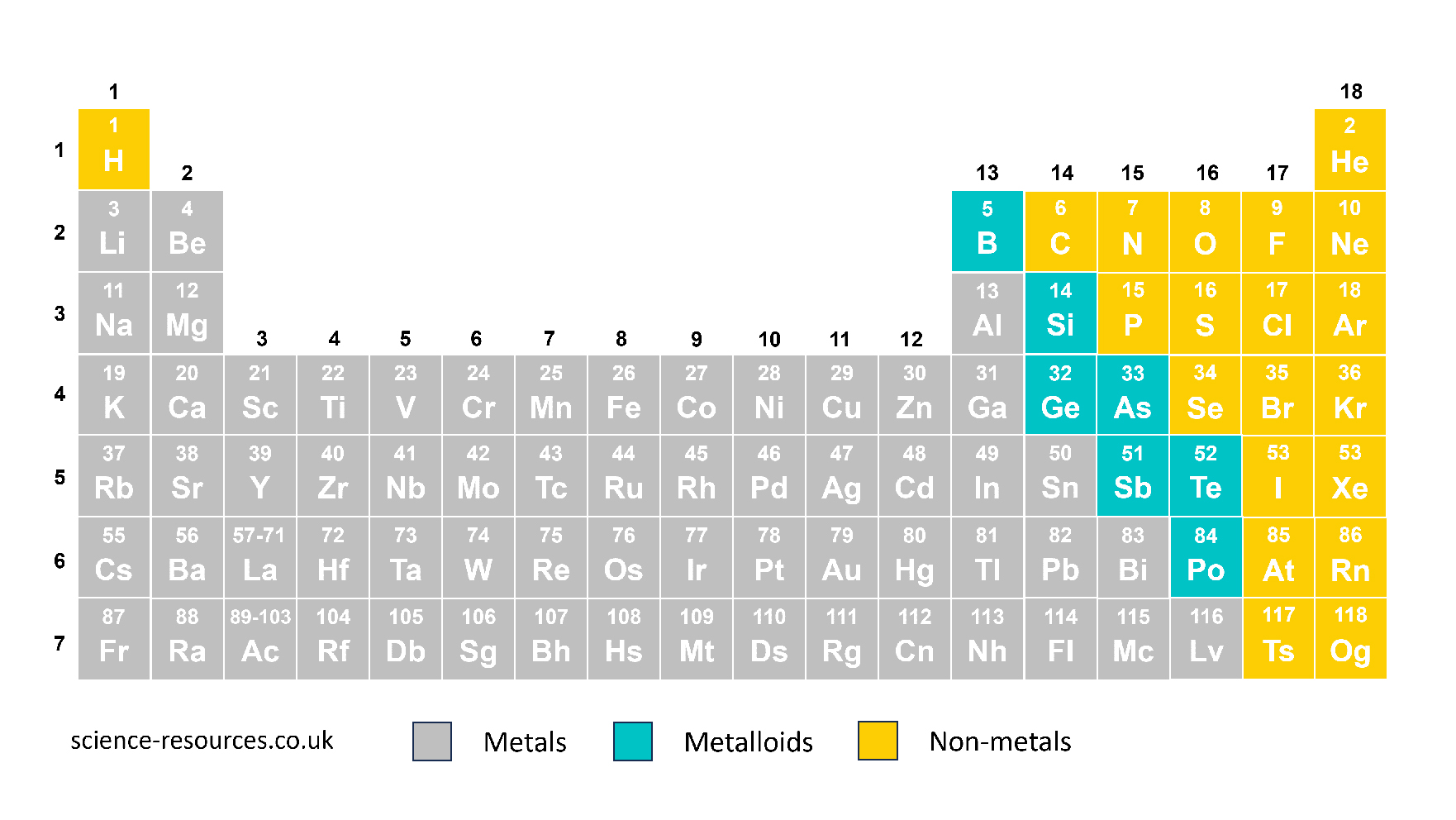
Compounds and molecules The atoms of the elements are joined together by chemical bonds in a fixed ratio of atoms. H2O = Two atoms of hydrogen, one atom of oxygen = 3 atoms in total Molecules Water molecule
Compounds
A compound is a substance that has atoms of two or more different elements.
The formula of a compound shows how many atoms of each element are in the compound.
The formula is made of symbols and numbers.
The numbers are written as subscript, which means they are smaller and lower than the symbol. For example, the formula for water is H2O.
For example:
CO2 = One atom of carbon, two atoms of oxygen = 3 atoms in total
Most molecules are made of two or more non-metal atoms bonded together. For example: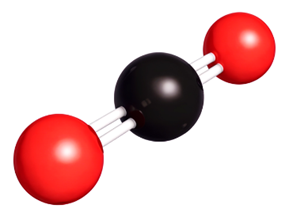
Test your ability to identify chemical symbols and understand how formulae are written. 1. Which of the following is the correct way to write the chemical symbol for Magnesium? 2. What is the correct chemical symbol for the element Potassium? 3. In the chemical formula CO₂, what does the number '2' represent? 4. When naming a compound like Sodium Chloride, why does the ending change to '-ide'? 5. Which of the following represents the correct formula for Magnesium Oxide? Summary: 🧪 Knowledge Check: Symbols and Formulae
Check Your Answers
2. K (Potassium uses its Latin-derived symbol).
3. Two atoms of Oxygen (The small number follows the symbol it refers to).
4. To show it is a non-metal in a compound (Compounds of a metal and non-metal usually end in -ide).
5. MgO (Correct symbols for Magnesium and Oxygen combined).