Changes of state
Changes of state
An ice cube is a solid form of water. When it is heated, it changes its state from solid to liquid. This process is called melting. If the liquid water is heated more, it changes its state from liquid to gas. This process is called boiling. Changes of state can also happen in reverse if a substance is cooled down. For example, if gas is cooled down, it can change its state from gas to liquid. This process is called condensation. If liquid is cooled down, it can change its state from liquid to solid. This process is called freezing.
How water changes state Arrangement of particles Solid Liquid Gas
This diagram shows the process of water changing state.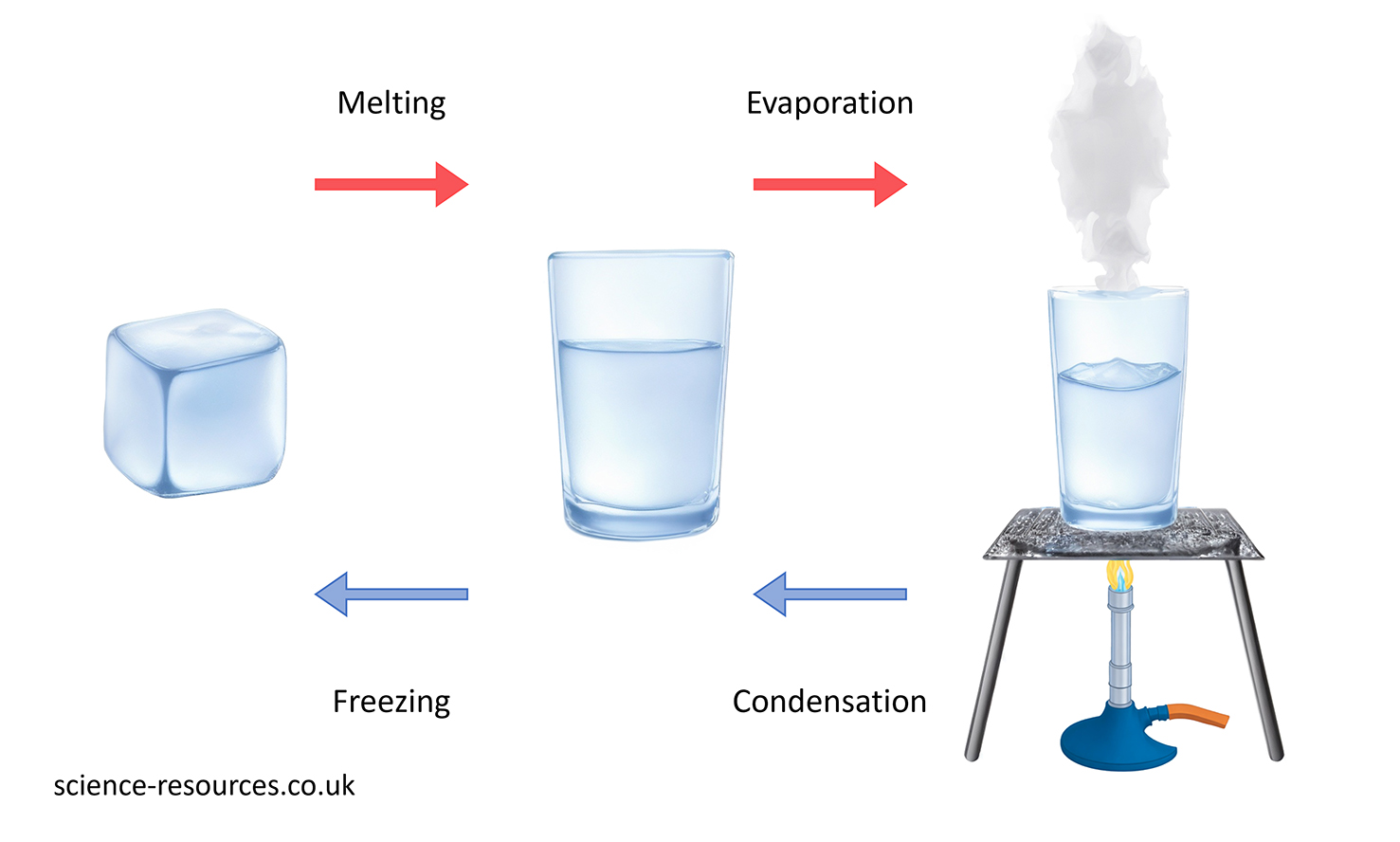
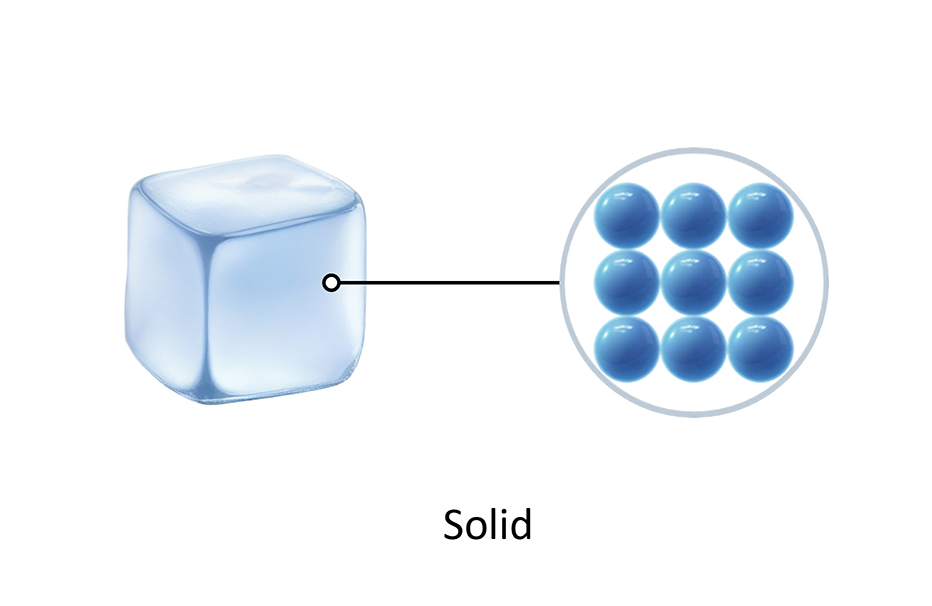
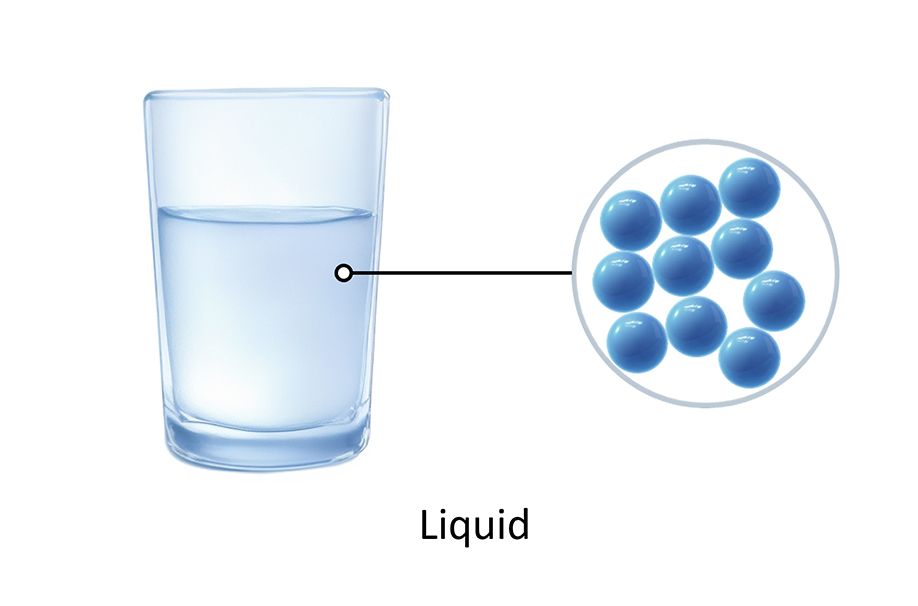
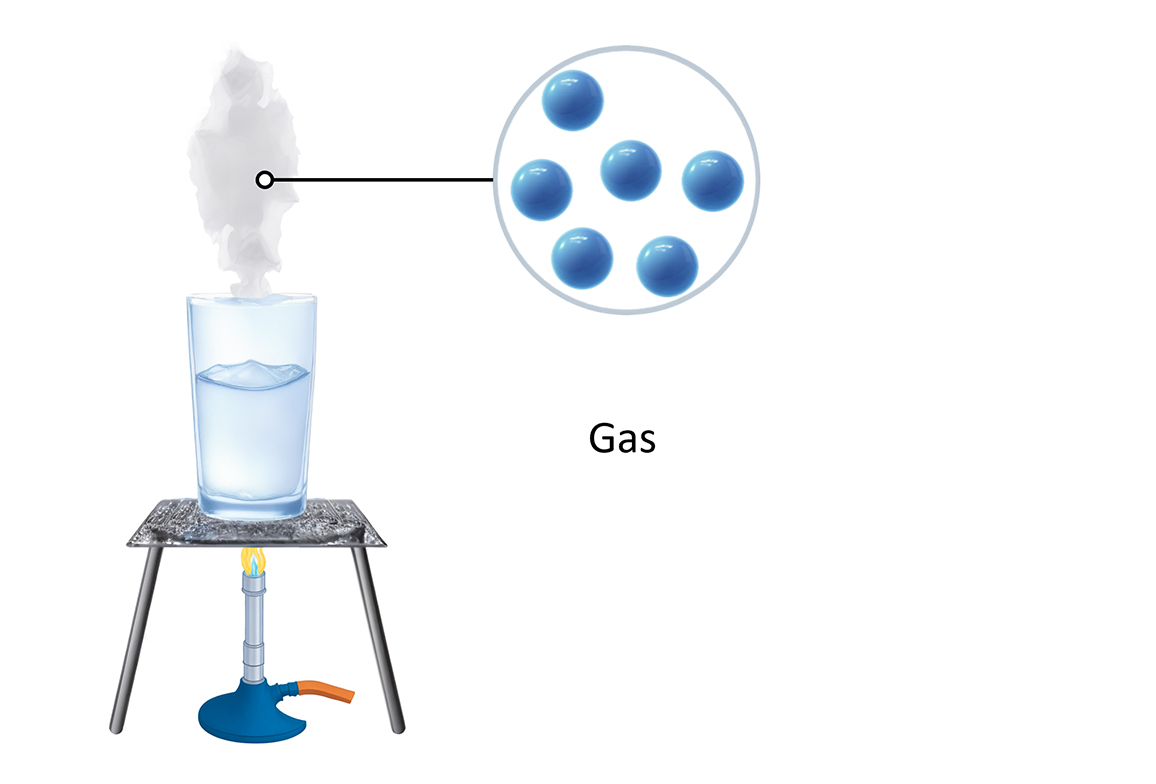
Test your knowledge on how matter changes from one state to another. 1. Which process describes a liquid turning into a solid? 2. What happens to the kinetic energy of particles when a substance is heated? 3. Which of the following is an example of a solid turning directly into a gas? 4. Why is a change of state described as a 'physical change'? 5. During condensation, what happens to the particles in a gas? Summary: 🧪 Knowledge Check: Changes of State
Click to Reveal Answers
2. Gain kinetic energy and move faster (Heating adds energy to the particles).
3. Subliming (Dry ice is a common example of this process).
4. No new substances are made (The substance stays the same, just in a different form).
5. Lose energy and get closer together (Cooling causes gas particles to form a liquid).