What is carbon dioxide?
Structure and properties
Carbon dioxide is a gas that you can’t see or smell at room temperature. It has one carbon atom and two oxygen atoms in each molecule, so its chemical formula is CO2.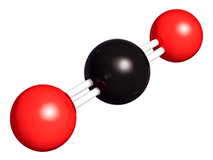
CO2 in the atmosphere Carbon dioxide is a gas that goes in and out of the air. Some things that add carbon dioxide to the air are cars that burn fuel and living things that breathe. Some things that take carbon dioxide from the air are plants that make food and water that dissolves it. Carbon dioxide is a small part of the air, but it is important because it keeps the Earth warm. It does this by trapping heat from the sun. But too much carbon dioxide can make the Earth too warm. This is called global warming, and it can change the weather in bad ways. For example, it can make some places very dry and some storms very strong. This is why we need to be careful about how much carbon dioxide we put in the air.
Test your understanding of the properties, testing, and environmental impact of carbon dioxide based on the webpage. 1. Which observation indicates a positive result when testing for carbon dioxide with limewater? 2. What is the name of the white precipitate formed during the limewater test? 3. How does the density of carbon dioxide compare to the density of air? 4. What is the environmental consequence of increasing carbon dioxide levels in the atmosphere? 5. Which of these is a physical property of carbon dioxide gas? Summary: 🧪 Knowledge Check: Carbon Dioxide
Click to Reveal Answers
2. Calcium carbonate (The insoluble solid that makes the liquid look cloudy).
3. It is denser than air (This allows it to be poured or collected in jars).
4. It enhances the greenhouse effect (Trapping more heat and causing global warming).
5. It is a colourless and odourless gas (It cannot be seen or smelled).