Heating and cooling
Hot vs. cold objects Example The cup of coffee has a starting temperature of 90 °C and cools to a temperature of 40 °C. Energy has moved to the surroundings. Thermal energy store Thermal equilibrium
The warmer an object, the more energy it has in its thermal energy store.
The average speed of particles in a warm substance is higher than in a cool substance.
Temperature is the measure of how hot a substance is. Temperature is usually measured in degrees Celsius (°C) using a thermometer.
Temperature depends on the average speed of the particles in a substance.
A hot cup of coffee cools down
Temperature
Warm coffee has more energy in its thermal energy store than cool coffee. As the coffee cooled, the average speed of the particles in the coffee decreased.
This energy didn’t vanish - it moved to the surroundings.
Energy has moved from the thermal energy store of the coffee to the thermal energy store of the surroundings.
When two objects have different temperatures, energy moves by heating from the warmer object to the colder one.
The bigger difference in temperature, the quicker the energy is transferred.
When the objects have the same temperature as the surrounding environment, the energy movement will stop.
When the objects share the same temperature, they are in thermal equilibrium.
Energy transfer Radiation All objects emit energy to their surroundings by infrared radiation. The warmer the object, the more infrared radiation it gives off. Conduction Convection
Energy is transferred by heating due to:
Infrared radiation is a kind of electromagnetic wave. Unlike conduction and convection, there are no particles involved. This means that energy can be moved by radiation even when there are no particles, like the emptiness of space.
Radiation is the reason we feel the heat of the Sun.
Conduction happens when energy is transferred by the vibrating of particles in a substance. The energy is transferred from a warmer area to a colder area.
Conduction occurs quickest in solids because the particles are near each other.
Materials that transfer energy easily from a warm area to a cold area by conduction are named conductors. Metals are very effective conductors.
Materials that do not transfer energy easily from a warm area to a cold area are named insulators. Air and plastics are good insulators.
The best insulator is a vacuum. Conduction cannot happen if there are no particles.
Convection happens in liquids and gases, because the particles are far apart and are free to move randomly. Although the particles in liquids are closer together than gases, they are still able to move and flow.
Near the heat source, particles acquire thermal energy, making them vibrate and move faster. This results in the particles spreading apart, making the liquid or gas less dense.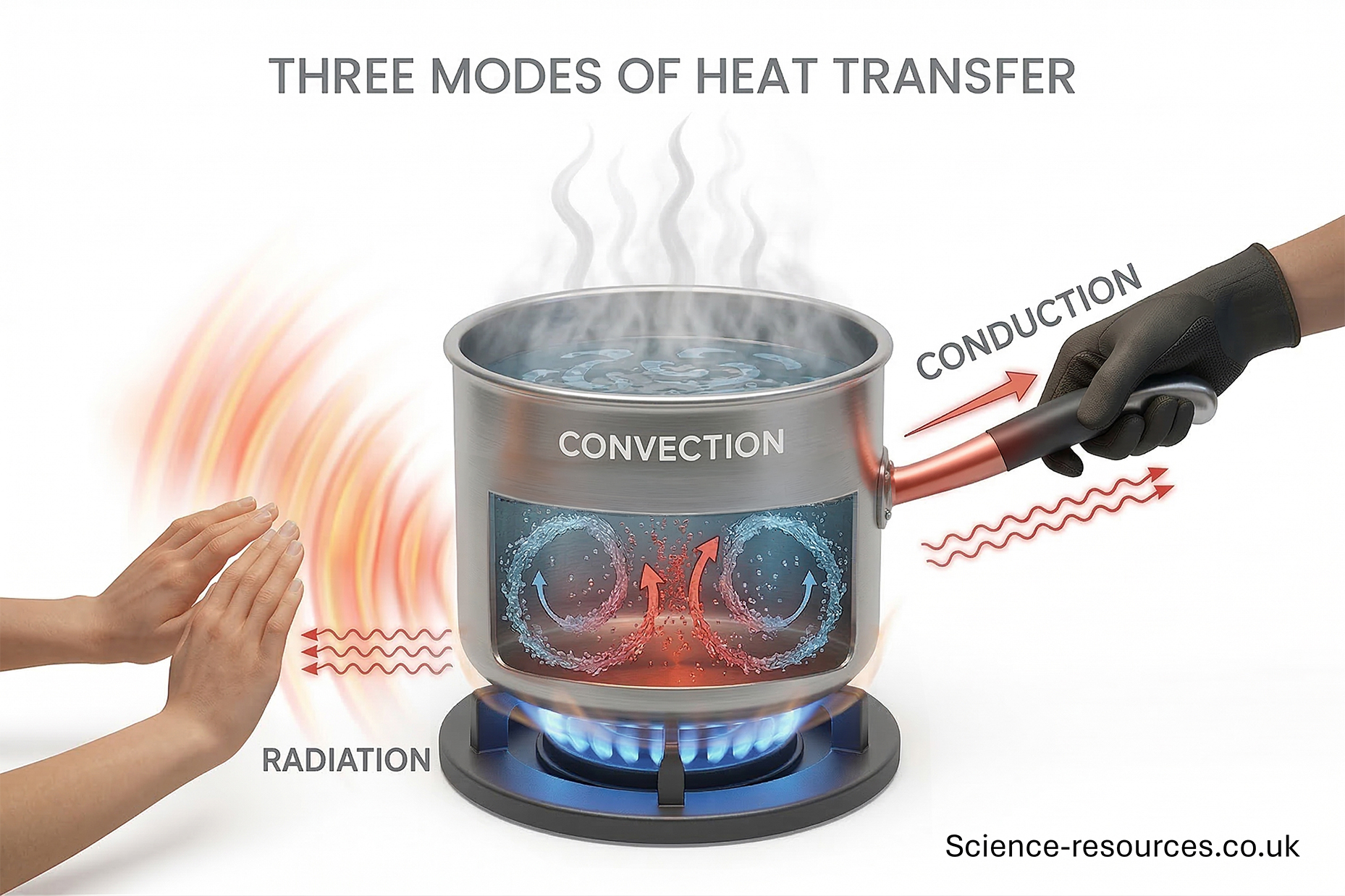 Three modes of heat transfer
Three modes of heat transfer
Test your understanding of how thermal energy moves through different materials and states of matter. 1. Which process involves the transfer of thermal energy through a solid by the vibration of particles? 2. In a convection current, why does a heated liquid or gas rise? 3. Which method of heat transfer can travel through the vacuum of empty space? 4. Why is wood often used for the handles of metal saucepans? 5. What happens to the particles in a liquid as it is cooled? Summary: 🔥 Knowledge Check: Heating and Cooling
Click to Reveal Answers
2. Because it becomes less dense (Expansion causes the heated fluid to rise).
3. Radiation (Infrared waves do not need particles to travel).
4. Because it is a good insulator (It prevents thermal energy from reaching your hand).
5. They lose energy and move more slowly (Cooling is the removal of thermal energy).