Changes of state
Change of state
Solids, liquids and gases are different states of matter that many substances can be in.
The state of a substance can be changed by heating it up or cooling it down. There are four main ways that a substance can change its state:
How water changes state There are four main changes of state: melting, freezing, evaporating and condensing Sublimation Conservation of mass
This diagram shows the process of water changing state.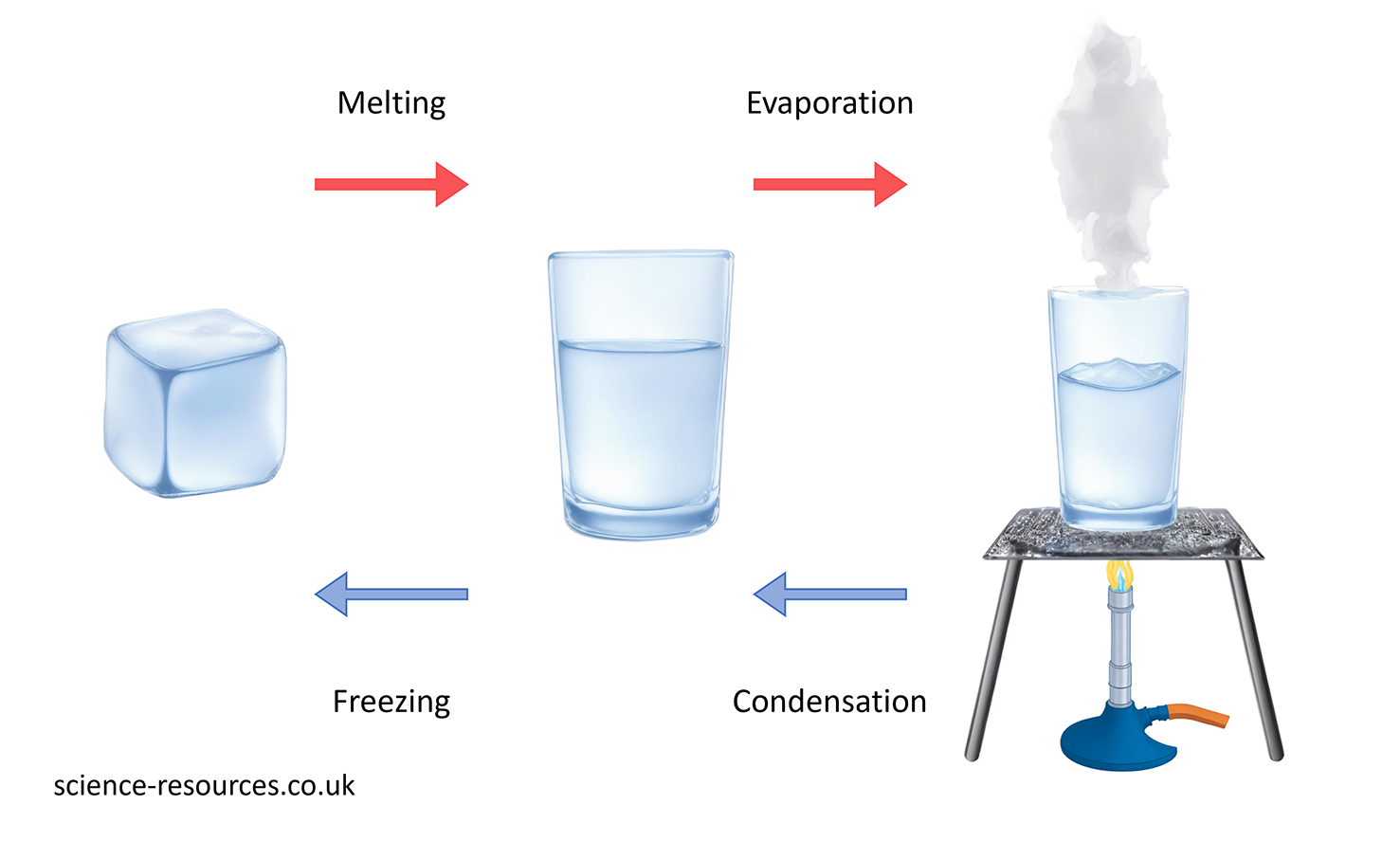
Some substances can change directly from solid to gas, or from gas to solid, without going through the liquid stage. This is called sublimation. Solid carbon dioxide ('dry ice') and iodine can sublime.
Mass does not change when a substance changes state. Only how the particles are arranged, how close they are, and how they move change. This is called the conservation of mass.
For example, 20 g of water boils to form 20 g of steam or freezes to form 20 g of ice. There is change of state, but no change of mass.
Test your knowledge of how substances transition between solids, liquids, and gases. 1. What is the name of the process when a liquid turns into a gas at a specific, fixed temperature? 2. Which change of state describes a gas cooling down to become a liquid? 3. True or False: Evaporation only takes place at the surface of a liquid. 4. What happens to the temperature of a substance while it is melting? 5. Sublimation is a special change of state where: Summary: 🔬 Knowledge Check: Changes of State
Click to Reveal Answers
2. Condensation (The process of gas turning back to liquid).
3. True (Unlike boiling, evaporation only occurs at the surface).
4. It stays the same (Temperature remains constant during any change of state).
5. A solid turns directly into a gas (Without passing through the liquid phase).