States of matter
Solids A particle model of a solid (Note: Particles are all touching) A particle model of a liquid (Note: The particles in a liquid are closely spaced and randomly arranged.)
Some examples of solid materials are paper, bricks, wood, metal and ice.
The particles in a solid are held together by strong forces that keep them in fixed places, in a regular pattern. The particles are very close and only shake around these fixed places. They cannot move around.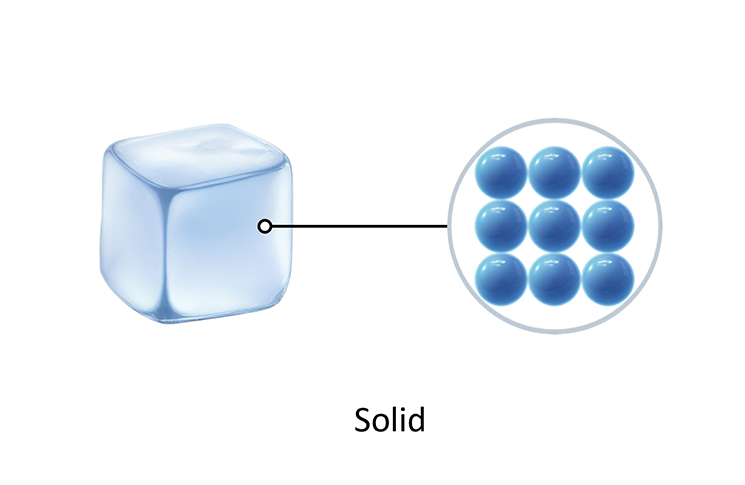
Liquids
Water, oil and fruit juice are some common examples of liquids. The particles in a liquid are not in fixed places because the forces between them are weaker than in a solid.
The particles in a liquid are still close together, but they are randomly arranged. They can vibrate and also move around.
Liquids do not have a fixed shape and can flow because the particles can move and vibrate. This means that a liquid will fit the shape of the bottom of its container. Like a solid, liquids cannot usually be squeezed because there is hardly any space between the particles.
The particles in a liquid should be drawn as close together with very few gaps, in a random pattern.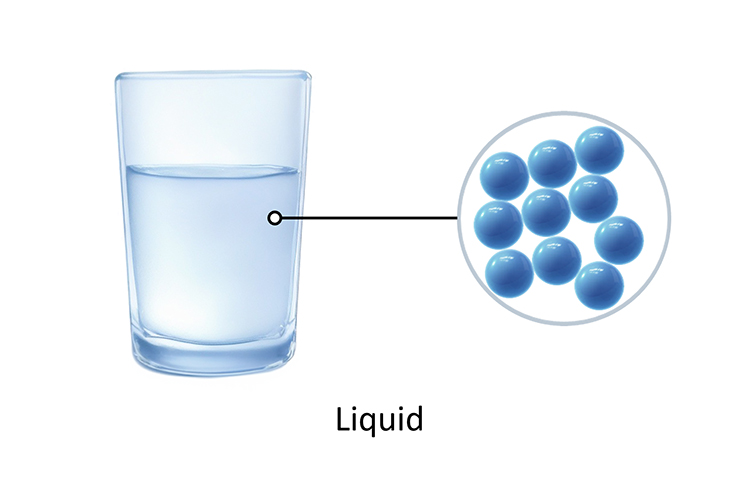
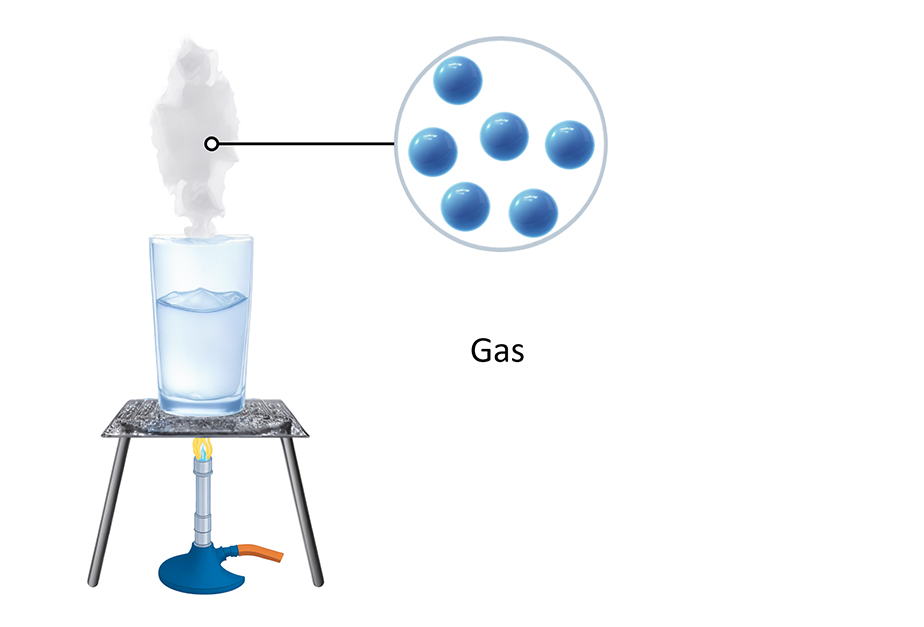
Gases The particles in a gas are so energetic that they can break free from the forces that try to keep them together. This makes the particles in a gas spread out and move in any direction. The particles in a gas do not shake - they move fast in straight lines, randomly. This means they can hit each other and the walls of any container they are in. Gases do not have a fixed shape because the particles can move freely in all directions. They can fill up their container and flow to take its shape. Gases can also be squeezed easily because there is a lot of space between the particles. The particles in a gas should be drawn as far apart and in a random pattern. Below is a table that compares the three states of matter. Solid Liquid Gas Arrangement of particles Regular arrangement Random arrangement Random arrangement Movement of particles Vibrate around a fixed position Move around each other Moves quickly in all directions Closeness of particles Very close together Close together with no regular arrangement Very spaced out
Some examples of gases are:
State
Summary: