What is water?
See also: What is water made of? Water molecule
Water is made of molecules. Each molecule has two hydrogen (H) atoms and one oxygen (O) atom.
This means that water’s chemical formula is H2O.
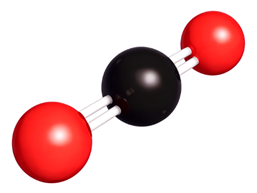
What makes water special? Why is water important? Humans need to drink enough water to replace what we lose though sweat, urine, and faeces.
Water is the most abundant substance on Earth’s surface. It covers more than 70% of the planet in oceans, rivers, lakes and glaciers.
Water has some unique properties:
Water is vital to all living things on Earth. Plants use water in photosynthesis to make their food.
About 60% of the human body is water and it does many things, such as:
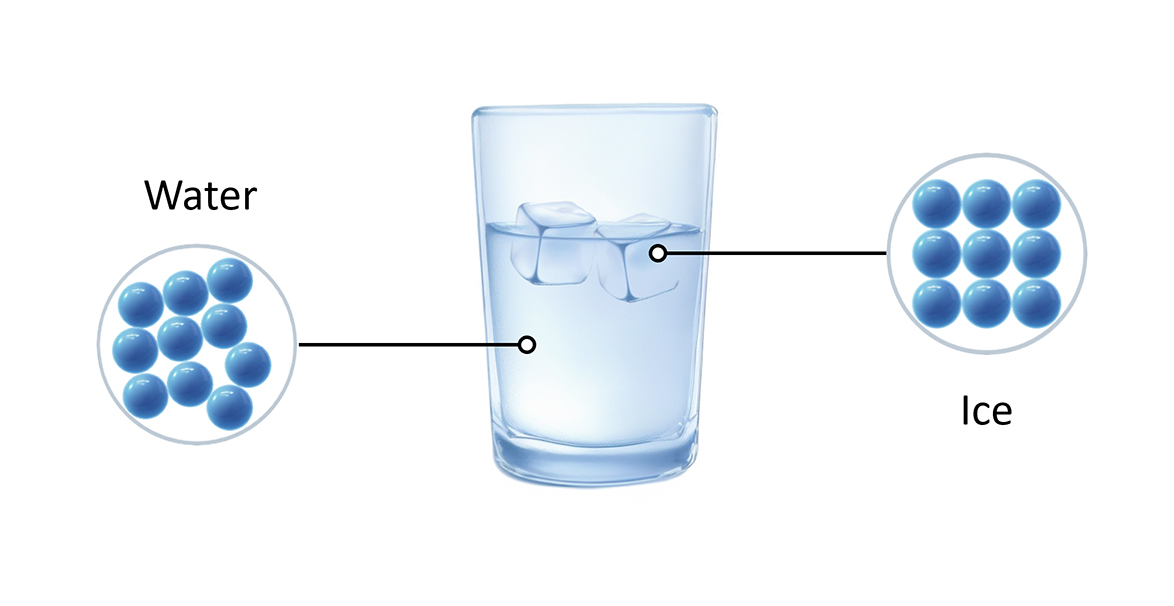
Summary: Test your understanding of how water particles behave as a solid, liquid, and gas. 1. In which state are the water particles arranged in a regular, fixed pattern? 2. What is the process called when liquid water is cooled and turns into ice? 3. How do particles behave in liquid water? 4. Which state of water takes the shape of its container but has a fixed volume? 5. What must be added to water to make it change from a liquid to a gas? Q1: What is water made of? A1: Water is made of molecules, each consisting of two hydrogen (H) atoms and one oxygen (O) atom, giving it the chemical formula H2O. Q2: What makes water special? A2: Water has unique properties: it can be solid, liquid, or gas at normal Earth temperatures, is most dense at 4°C, dissolves many substances, and has high surface tension. Q3: Why is water important? A3: Water is vital for all living things. It is used in photosynthesis, makes up about 60% of the human body, and helps dissolve nutrients, regulate temperature, remove waste, and protect tissues. Q4: How much of Earth’s surface is covered by water? A4: Water covers more than 70% of Earth’s surface in oceans, rivers, lakes, and glaciers. Q5: What are the three states of water? A5: Water can exist as a solid (ice), liquid (water), or gas (vapor). Q6: At what temperature is water most dense? A6: Water is most dense at 4°C. Q7: How does water benefit the human body? A7: Water dissolves nutrients, regulates body temperature, removes waste, and protects tissues, joints, and the spinal cord. Q8: Why do humans need to drink water? A8: Humans need to drink water to replace what is lost through sweat, urine, and feces. Q9: What role does water play in photosynthesis? A9: Water is used by plants in photosynthesis to make their food. Q10: What substances can water dissolve? A10: Water can dissolve many substances, including salts and proteins.
Next:
Changes of state
Solids, liquids, gases
🔬 Knowledge Check: The States of Water
Click to Reveal Answers
2. Freezing (The change from liquid to solid).
3. Close together but can move past each other (This allows liquids to flow).
4. Liquid water (Liquids flow to fit a container but do not expand like gases).
5. Heat energy (Energy is needed to break the bonds between liquid particles).
What is water FAQ