Chromatography
Separating dissolved solids using chromatography Chromatography works because some of the colours dissolve more easily in water than others. Colours that dissolve more easily travel further up the paper. A chromatogram with different samples of ink.
Chromatography is a method of separating mixtures of substances that can dissolve in water.
This is especially easy when the dissolved substances have different colours. An ink or food colouring is usually made from more than one colour. Chromatography can be used to find out the colours in ink or food colouring.
There are two ways to do chromatography:
Standard chromatography
The photo shows an example of a paper with different colours from standard chromatography.
Four samples of ink were used. Looking at the photo from left to right, the first sample on the left is a mixture of colours (yellow and orange). The other three samples are single colours (blue, pink and yellow).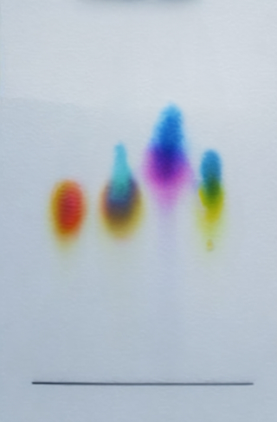
Radial chromatography This is done by putting water on a spot of ink in the middle of a piece of filter paper. The colours spread out as they move away from the middle, rather than moving up as in standard chromatography. Comparing chromatograms
Another way to separate colours in an ink is radial chromatography.
Chromatography is often used to see if an unknown mixture has the same colours as pure pigments. This helps scientists to identify the colours in the mixture. Pure pigments that are used for this purpose are called standard reference pigments.
The piece of paper that is left after a chromatography experiment is called a chromatogram. A chromatogram shows the colours that were in the mixture.
Test your understanding of how chromatography works and how to interpret the results. 1. In paper chromatography, what is the 'mobile phase'? 2. Why should the start line be drawn in pencil rather than ink? 3. If a substance produces three distinct spots on a chromatogram, what does this tell you? 4. How is the Rf value of a substance calculated? 5. A substance that is highly soluble in the mobile phase will: Summary: 🧪 Knowledge Check: Chromatography
Click to Reveal Answers
2. Pencil lead is insoluble (This ensures the start line stays fixed).
3. A mixture of at least three components (Each spot represents a different substance).
4. Distance moved by substance ÷ Distance moved by solvent (This ratio is always between 0 and 1).
5. Move a long distance up the paper (Greater solubility results in a higher Rf value).